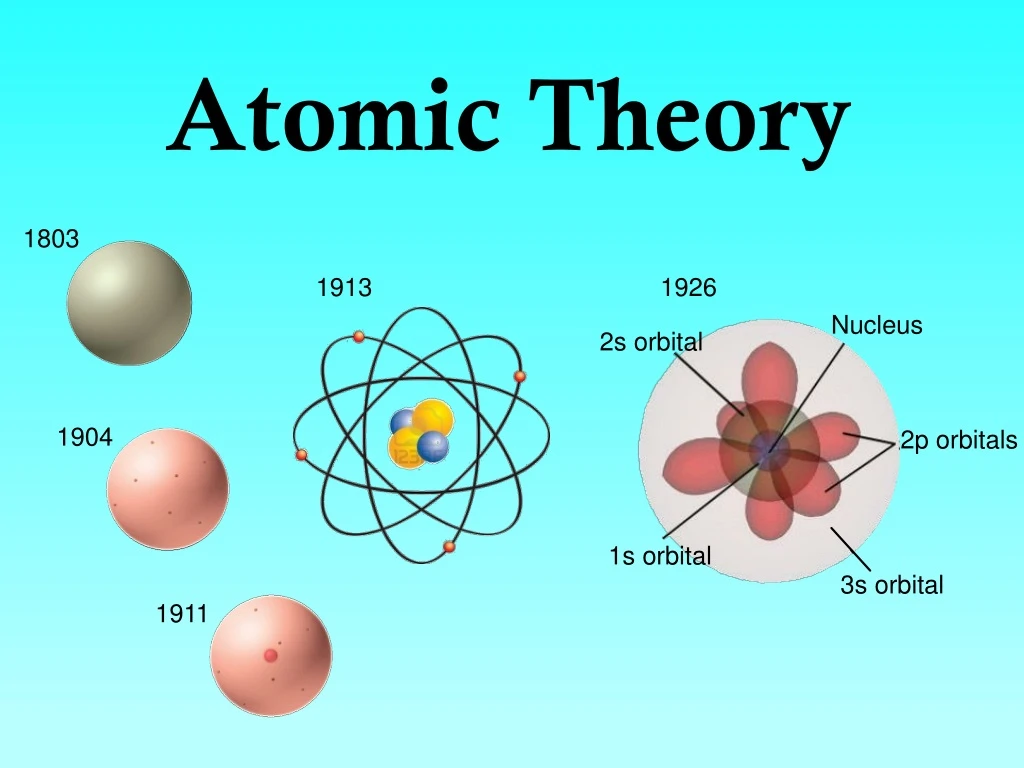
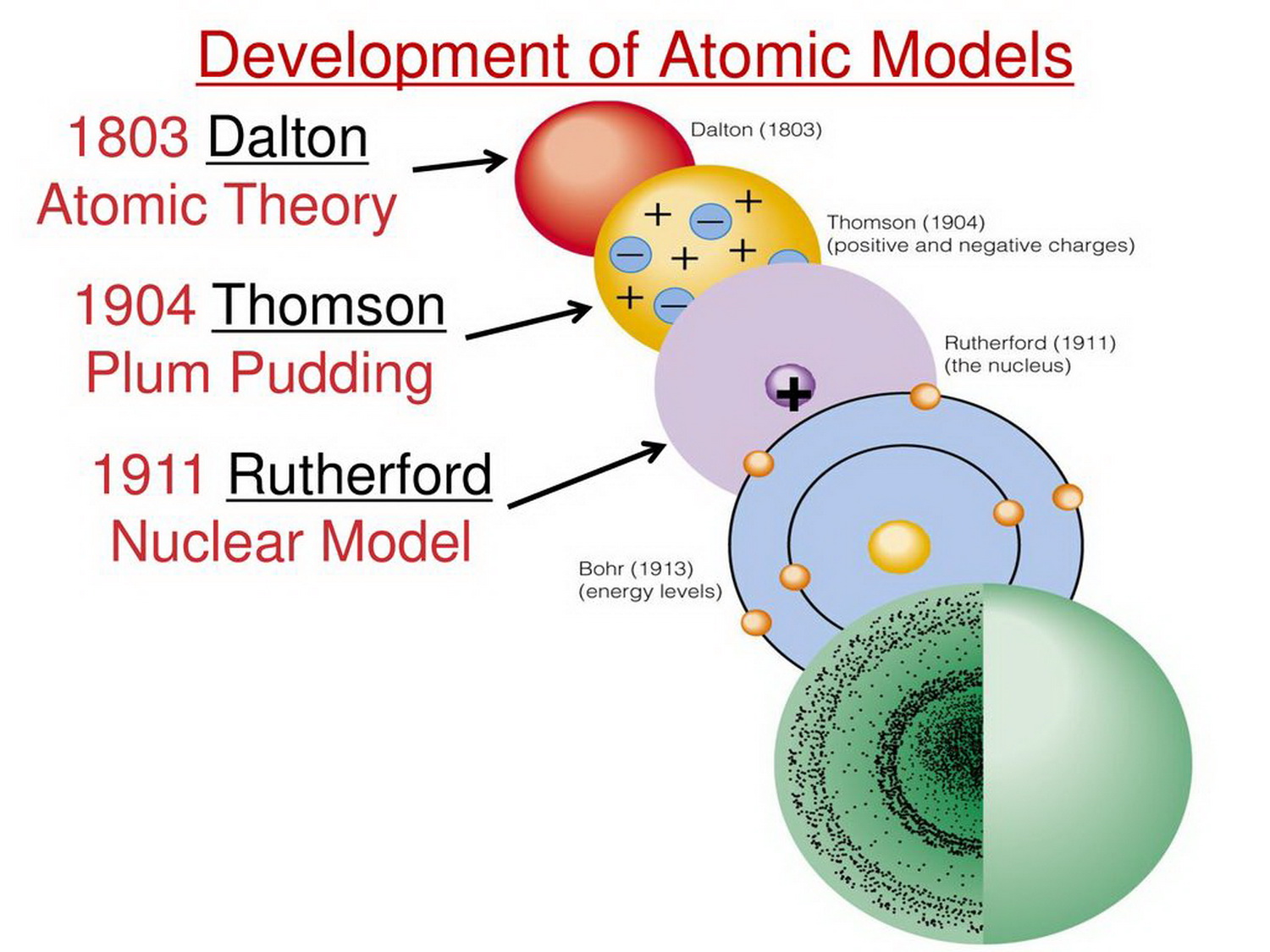
 This means that the negative charge on an electron perfectly balances the positive charge on the proton. Negative and positive charges of equal magnitude cancel each other out. Neutral atoms have the same number of electrons as they have protons, so their overall charge is zero. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite numbers through empty space until stopped. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative. The work of Ernest Rutherford, Niels Bohr and others, suggested that an atom consists of a central core, the nucleus, surrounded by one or more electrons, arranged in energy levels each of which can hold some specific number of electrons. Mass of electron 1.602×1019C× 1kg 1.759×1011C 9.107×1031kg Mass of electron 1.602 × 10 19 C × 1 kg 1.759 × 10 11 C 9.107 × 10 31 kg. We will discuss isotopes and their symbols more in section 4.5 of this chapter. By 1913, the main features of the modern atomic theory had been worked out. Atoms of the same element (i.e., atoms with the same number of protons) with different numbers of neutrons are called isotopes. The sum of the number of protons and neutrons in the nucleus is called the mass number of the isotope. Each element has its own characteristic atomic number.Ītoms of the same element can have different numbers of neutrons, however. Thus, hydrogen has an atomic number of 1, while iron has an atomic number of 26. The number of protons in an atom is the atomic number of the element (Z). This number of protons is so important to the identity of an atom that it is called the atomic number. All atoms of hydrogen have one and only one proton in the nucleus all atoms of iron have 26 protons in the nucleus. 
What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
